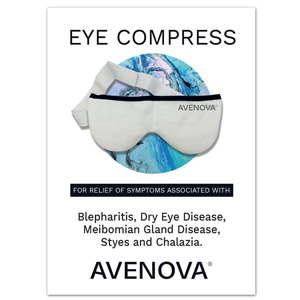
EMERYVILLE, Calif. (March 31, 2021) – NovaBay® Pharmaceuticals, Inc. (NYSE American: NBY), a pharmaceutical company focusing on commercializing Avenova for the eye care market, announces the availability of its new Warm Eye Compress on Amazon.com and on Avenova.com. This new, reusable heated eye compress is designed to relieve the symptoms associated with blepharitis, dry eye disease, meibomian gland disease, styes and chalazion, and is a companion product to Avenova, NovaBay’s premier antimicrobial lid and lash spray.
 “Most people who suffer from dry eye are unable to produce adequate lubrication for their eyes, either due to insufficient tear production or poor-quality tears,” said Justin Hall, President and CEO of NovaBay Pharmaceuticals. “Avenova Warm Eye Compress helps to hydrate and lubricate eyes to efficiently expel bacteria and debris, making eyes feel refreshed and rejuvenated. When used together with our Avenova lid and lash spray – which is clinically proven to treat the underlying cause of bacterial dry eye, rather than just the symptoms – the eyes will start to restore the natural defenses against tear evaporation.”
“Most people who suffer from dry eye are unable to produce adequate lubrication for their eyes, either due to insufficient tear production or poor-quality tears,” said Justin Hall, President and CEO of NovaBay Pharmaceuticals. “Avenova Warm Eye Compress helps to hydrate and lubricate eyes to efficiently expel bacteria and debris, making eyes feel refreshed and rejuvenated. When used together with our Avenova lid and lash spray – which is clinically proven to treat the underlying cause of bacterial dry eye, rather than just the symptoms – the eyes will start to restore the natural defenses against tear evaporation.”
The Avenova Warm Eye Compress stimulates the meibomian glands to produce more tears, thereby clearing these glands and allowing natural oils to flow back onto the eye, as well as preventing tears from evaporating too quickly. The compress also improves blood circulation and helps to relax the eye, which may increase comfort.
To enjoy the many benefits of the Avenova Warm Eye Compress, simply heat it in a microwave oven for 15-20 seconds and place it over closed eyelids for up to 15-20 minutes or as directed by a physician. Then gently wipe the skin surrounding your eyes with a wet cloth. The compress should be left to air dry or can be hand washed, and will usually be ready for reuse within two hours. Currently in stock on both the Amazon and the Avenova websites, the Avenova Warm Eye Compress is available for $17.50 and shipping is free on Avenova.com.
About NovaBay Pharmaceuticals, Inc.: Going Beyond Antibiotics®
NovaBay Pharmaceuticals, Inc. is a biopharmaceutical company focusing on high-quality, differentiated, anti-infective consumer products: Avenova®, the premier antimicrobial lid and lash spray, CelleRx® Clinical Reset™, a breakthrough product in the beauty category, and NeutroPhase® Skin and Wound Cleanser for wound healing. NovaBay’s products are formulated with its patented, pure, stable, pharmaceutical-grade hypochlorous acid that replicates the antimicrobial chemicals used by white blood cells to fight infection. NovaBay’s hypochlorous acid products do not cause stinging or irritation, are non-toxic and non-sensitizing, making them completely safe for regular use. Avenova is the only commercial hypochlorous acid lid and lash spray product clinically proven to reduce bacterial load on ocular skin surfaces, thus effectively addressing the underlying cause of bacterial dry eye.
Forward-Looking Statements
Except for historical information herein, matters set forth in this press release are forward-looking within the meaning of the “safe harbor” provisions of the Private Securities Litigation Reform Act of 1995, including statements about the commercial progress and future financial performance of NovaBay Pharmaceuticals, Inc. This release contains forward-looking statements that are based upon management’s current expectations, assumptions, estimates, projections and beliefs. These statements include, but are not limited to, statements regarding our current product offerings, potential future product offerings, and any future revenue that may result from selling these products, as well as generally the Company’s expected future financial results. These statements involve known and unknown risks, uncertainties and other factors that may cause actual results or achievements to be materially different and adverse from those expressed in or implied by the forward-looking statements. Factors that might cause or contribute to such differences include, but are not limited to, risks and uncertainties relating to the size of the potential market for our products, the possibility that the available market for the Company’s products will not be as large as expected, the Company’s products will not be able to penetrate one or more targeted markets, revenues will not be sufficient to meet the Company’s cash needs, the effect on sales and potential reputational damage resulting from decisions or actions taken by regulators, including Warning Letters issued by the FDA, and any other potential regulatory problems that may arise. Other risks relating to NovaBay’s business, including risks that could cause results to differ materially from those projected in the forward-looking statements in this press release, are detailed in NovaBay’s latest Form 10-Q/K filings with the Securities and Exchange Commission, especially under the heading “Risk Factors.” The forward-looking statements in this release speak only as of this date, and NovaBay disclaims any intent or obligation to revise or update publicly any forward-looking statement except as required by law.
Socialize and Stay informed on NovaBay’s progress
Like us on Facebook
Follow us on Twitter
Connect with NovaBay on LinkedIn
Visit NovaBay’s Website
Avenova Purchasing Information
For NovaBay Avenova purchasing information:
Please call 800-890-0329 or email sales@avenova.com
www.Avenova.com
CelleRx Clinical Reset Purchasing Information
For NovaBay CelleRx Clinical Reset purchasing information
Please call 877-CELLERX
www.CelleRx.com
NovaBay Contact
Justin Hall
Chief Executive Officer and General Counsel
510-899-8800
jhall@novabay.com
Investor Contact
LHA Investor Relations
Jody Cain
310-691-7100
jcain@lhai.com