• Net product revenue of $1.9 million, up 30% year-over-year
• 51% of Avenova® sales generated from Amazon.com, up from 35% in the fourth quarter
• Sales of KN95 masks near one million units
• Emergency Use Approval for COVID-19 test kits filed on April 17, 2020
Conference call begins at 4:30 p.m. Eastern time today
EMERYVILLE, Calif. (May 7, 2020) – NovaBay® Pharmaceuticals, Inc. (NYSE American: NBY) reports financial results for the three months ended March 31, 2020 and provides a business update.
“First quarter net product revenue of $1.9 million increased 30% over the prior year as we benefitted from higher Avenova unit sales and from initial KN95 masks sales,” said Justin Hall, President and CEO of NovaBay Pharmaceuticals. “Online sales for the first time accounted for more than half of all Avenova sales for the quarter, up from 35% for the fourth quarter of 2019. We are making significant progress with our strategy to diversify our product offerings and sales channels in an effort to offset changes in industrywide prescription reimbursement that have impacted Avenova sales in past years.
“During this economic slowdown, we tapped into our global healthcare supply network to secure in-demand items that we can offer at reasonable prices. We are nearing the milestone of selling one million KN95 masks, which includes several large orders sold at discounted pricing. Most of these sales occurred in April,” he added. “We also have submitted the fluorecare® rapid, point-of-care COVID-19 antibody test for Emergency Use Authorization (EUA) with the U.S. Food and Drug Administration (FDA) for sale to U.S. healthcare professionals, which follows our recently announced distribution agreement. While our business faces considerable uncertainty from the COVID-19 pandemic, the nimbleness of our organization enabled us to quickly add the masks to our existing product offerings and submit the test kits for FDA EUA approval. I’m pleased to report that our entire organization is prepared to support a return to full operations following the lifting of shelter-in-place restrictions.”
NovaBay also announced the appointment of Andy Jones as Chief Financial Officer and Treasurer effective May 4, 2020. “I am honored to be joining the team at such an exciting time in the company’s development,” said Mr. Jones. “Recent events, including the diversification of product offerings and sales channels, demonstrate the forward thinking nature of the NovaBay team. I look forward to using my experience to support these and future initiatives to achieve continued success.”
Because the regulatory landscape for the test kits has been changing so rapidly, NovaBay cannot assure a timeline for FDA review and/or clearance for commercial marketing under EUA, or if clearance will be granted at all.
First Quarter Financial Results
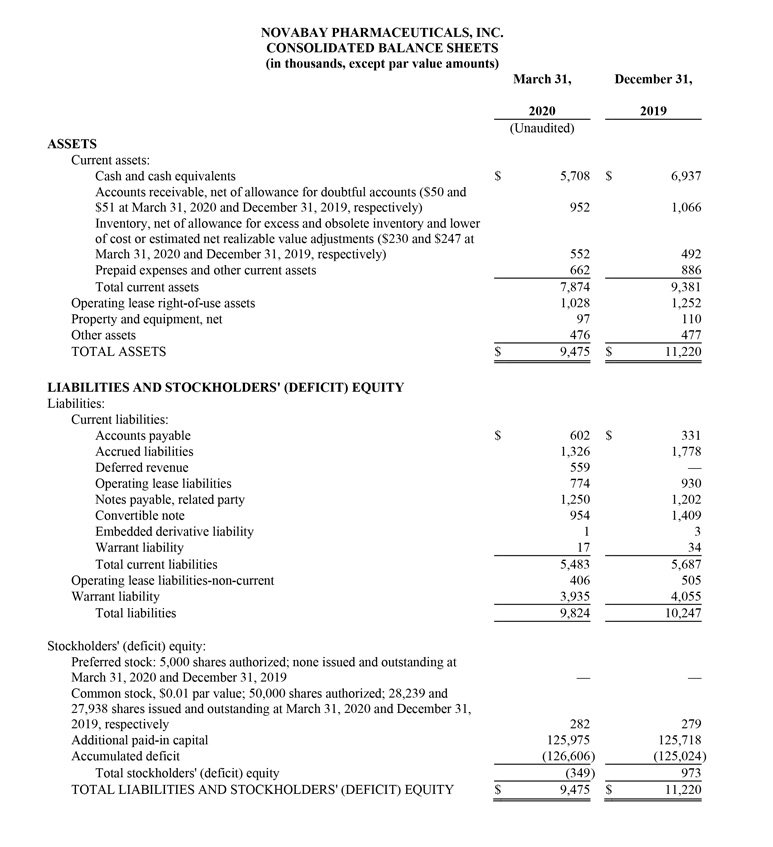
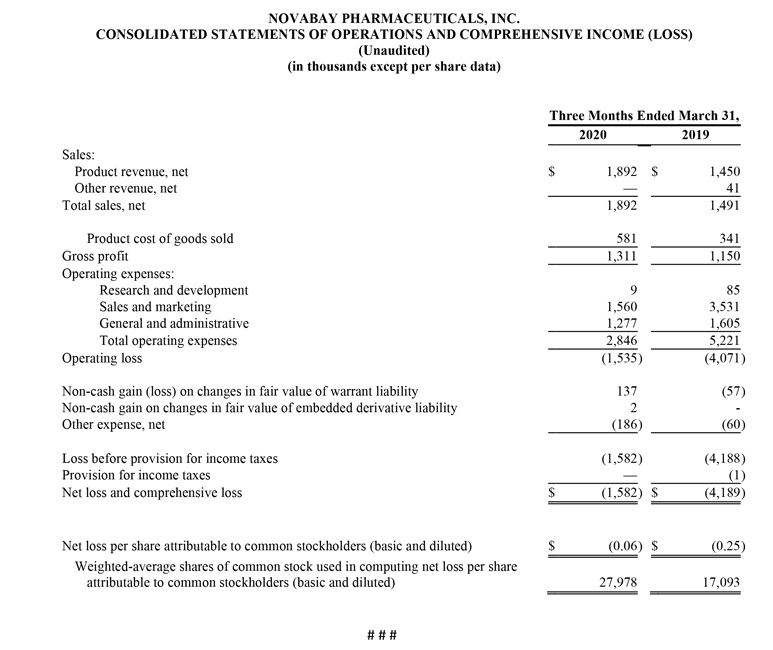
Net product revenue for the first quarter of 2020 was $1.9 million, a 30% increase from $1.5 million for the first quarter of 2019. The increase is due to higher Avenova unit sales, partially offset by a lower net selling price largely due to a decrease in insurance coverage by national payors. In response to this pricing dynamic, in June 2019 NovaBay launched Avenova sales on Amazon.com, which is helping to stabilize Avenova unit sales but at a lower net selling price. First quarter net sales also included $176,000 in sales of KN95 masks and $173,000 from a shipment of NeutroPhase® to NovaBay’s distribution partner China Pioneer Pharma Holdings. Gross margin on net product revenue was 69% for the first quarter of 2020, compared with 77% for the prior-year period, with the decrease due to lower Avenova gross-to-net pricing.
Operating expenses for the first quarter of 2020 were $2.8 million, a 45% decline from $5.2 million for the first quarter of 2019, reflecting the strategic shift in the U.S. commercial organization undertaken in March 2019 that included a major salesforce reduction, partially offset by an increase in direct marketing expenses focused on increasing sales of Avenova. Sales and marketing expenses for the first quarter of 2020 were $1.6 million, a 56% decline from $3.5 million for the first quarter of 2019 reflecting lower headcount, partially offset by an increase in direct marketing expenses. General and administrative (G&A) expenses for the first quarter of 2020 were $1.3 million, a 20% decline from $1.6 million for the first quarter of 2019 due to the departure of the Company’s interim CEO/CFO and reduction in headcount, both during the first quarter of 2019. Research and development (R&D) expenses for the first quarter of 2020 were $9,000, compared with $85,000 for the first quarter of 2019.
Operating loss for the first quarter of 2020 was $1.5 million, a 62% improvement from $4.1 million for the first quarter of 2019.
Non-cash gain on the change of fair value of warrant liability for the first quarter of 2020 was $137,000, compared with a non-cash loss of $57,000 for the first quarter of 2019.
Other expense, consisting primarily of interest expense, was $186,000 for the first quarter of 2020 compared with $60,000 for the first quarter of 2019. The increase was a result of debt issued during the first quarter of 2019, which was outstanding for the entire first quarter of 2020.
The net loss attributed to common stockholders for the first quarter of 2020 was $1.6 million, or $0.06 per share, compared with a net loss for the first quarter of 2019 of $4.2 million, or $0.25 per share.
NovaBay reported cash and cash equivalents of $5.7 million as of March 31, 2020, compared with $6.9 million as of December 31, 2019.
Conference Call
NovaBay management will host an investment community conference call today beginning at 4:30 p.m. Eastern time (1:30 p.m. Pacific time) to discuss the Company’s financial and operational results and to answer questions. Shareholders and other interested parties may participate in the conference call by dialing 800-608-8202 from within the U.S. or 702-495-1913 from outside the U.S., with the conference identification number 7080709.
A live webcast of the call will be available at https://novabay.com/investors/events and will be archived for 90 days. A replay of the call will be available beginning two hours after the call ends through 11:59 p.m. Eastern time May 28, 2020 by dialing 855-859-2056 from within the U.S. or 404-537-3406 from outside the U.S., and entering the conference identification number 7080709.
About NovaBay Pharmaceuticals, Inc.: Going Beyond Antibiotics®
NovaBay Pharmaceuticals, Inc. is a biopharmaceutical company focusing on commercializing and developing its non-antibiotic anti-infective products to address the unmet therapeutic needs of the global, topical anti-infective market with its two distinct product categories: the NEUTROX® family of products and the AGANOCIDE® compounds. The Neutrox family of products includes AVENOVA® for the eye care market, CELLERX® for the aesthetic dermatology market and NEUTROPHASE® for the wound care market. The Aganocide compounds, still under development, have target applications in the dermatology and urology markets.
Forward-Looking Statements
Except for historical information herein, matters set forth in this press release are forward-looking within the meaning of the “safe harbor” provisions of the Private Securities Litigation Reform Act of 1995, including statements about the commercial progress and future financial performance of NovaBay Pharmaceuticals, Inc. This release contains forward-looking statements that are based upon management’s current expectations, assumptions, estimates, projections and beliefs. These statements include, but are not limited to, statements regarding our business strategies and current product offerings, potential future product offerings, possible regulatory clearance of any of our products or future products, and any future revenue that may result from selling these products, as well as generally the Company’s expected future financial results. These statements involve known and unknown risks, uncertainties and other factors that may cause actual results or achievements to be materially different and adverse from those expressed in or implied by the forward-looking statements. Factors that might cause or contribute to such differences include, but are not limited to, risks and uncertainties relating to the size of the potential market for our products, the possibility that the available market for the Company’s products will not be as large as expected, the Company’s products will not be able to penetrate one or more targeted markets, revenues will not be sufficient to meet the Company’s cash needs, and any potential regulatory problems. Other risks relating to NovaBay’s business, including risks that could cause results to differ materially from those projected in the forward-looking statements in this press release, are detailed in NovaBay’s latest Form 10-Q/K filings with the Securities and Exchange Commission, especially under the heading “Risk Factors.” The forward-looking statements in this release speak only as of this date, and NovaBay disclaims any intent or obligation to revise or update publicly any forward-looking statement except as required by law.
Socialize and Stay informed on NovaBay’s progress
Like us on Facebook
Follow us on Twitter
Connect with NovaBay on LinkedIn
Visit NovaBay’s Website
Avenova Purchasing Information
For NovaBay Avenova purchasing information:
Please call 800-890-0329 or email sales@avenova.com
www.Avenova.com
NovaBay Contact
Justin Hall
President and Chief Executive Officer
510-899-8800
jhall@novabay.com
Investor Contact
LHA Investor Relations
Jody Cain
310-691-7100
jcain@lhai.com